Project 1: New reactions of Ketenes
Example: (A) Catalytic asymmetric synthesis of bicyclic pyrazolidinones from ketenes 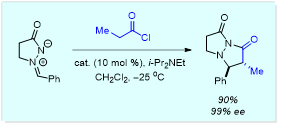
Pharmacologically interesting bicyclic pyrazolidinones have proven difficult to access through currently available technologies. We have recently developed a route that relies on the use of an alkaloid catalyst to affect the highly enantioselective (up to >99% ee) and diastereoselective (dr up to 27:1) synthesis of the desired structural motif from in situ-generated ketenes and bench stable azomethine imines (Org. Lett. 2016, 4108). Future work will involve an evaluation of the scope of this process, e.g. do disubstituted ketenes work as substrates? These heterocyclic molecules offer potential for collaborations relating to the treatment of Alzheimer’s disease, and evaluation of derivatives for use as herbicides or pesticides. Another important direction of research will explore immobilization of alkaloid and phosphine catalysts upon magnetic nanoparticle and polystyrene supports as a means of enabling efficient catalyst recycling for this and related organocatalytic reactions.
A number of other ketene-centred projects are in various stages of development.
Project 2: Asymmetric synthesis of 5-membered ring systems from sulfoxonium salts
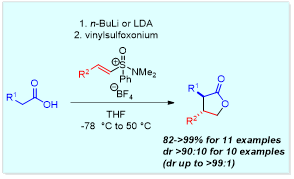 In 2013 we reported a diastereoselective synthesis of gamma-lactones, through interception of a sulfoxonium ylide-derived betaine intermediate by disubstituted ketenes (J. Org. Chem. 2013, 4587). In addition, we have recently determined that the reaction of lithium enediolates with vinyl sulfoxonium salts provides an efficient and highly diastereoselective route to gamma-lactones, favouring the trans-isomer (Org. Lett. 2015, 1735). An asymmetric variant, relying on the use of stoichiometric amounts of chiral amine (‘Koga tetraamine’) to interact with the organolithium intermediates, facilitated an enantioselective synthesis of 3,4-substituted gamma-lactones (J. Org. Chem. 2017, 606). Future work will focus on the development of a catalytic asymmetric variant, which would likely have greater appeal to synthetic organic chemists in industry.
In 2013 we reported a diastereoselective synthesis of gamma-lactones, through interception of a sulfoxonium ylide-derived betaine intermediate by disubstituted ketenes (J. Org. Chem. 2013, 4587). In addition, we have recently determined that the reaction of lithium enediolates with vinyl sulfoxonium salts provides an efficient and highly diastereoselective route to gamma-lactones, favouring the trans-isomer (Org. Lett. 2015, 1735). An asymmetric variant, relying on the use of stoichiometric amounts of chiral amine (‘Koga tetraamine’) to interact with the organolithium intermediates, facilitated an enantioselective synthesis of 3,4-substituted gamma-lactones (J. Org. Chem. 2017, 606). Future work will focus on the development of a catalytic asymmetric variant, which would likely have greater appeal to synthetic organic chemists in industry.
Project 3: Asymmetric synthesis of deoxypropionate and dipropionate derivatives
Ketene heterodimerization (cross-dimerization) had long been viewed as a difficult reaction to develop due to the difficulty of controlling regioselectivity, where multiple dimer regioisomers could potentially be formed. We recently disclosed the first catalytic asymmetric heterodimerization of ketenes (J. Am. Chem. Soc. 2012, 134, 2942 and J. Org. Chem. 2016, 7824). This reaction was catalyzed by an alkaloid nucleophile (a quinine or quinidine derivative) to give ketene heterodimer β-lactones in regioselective (only one heterodimer formed), enantioselective (21 Z-isomer examples ≥90% ee), and diastereoselective fashion (only the Z-isomer formed).
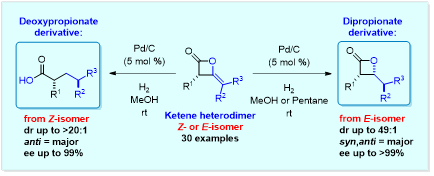
Our ultimate goal is to elaborate ketene heterodimers to biologically interesting complex molecules with great potential for medicinal applications. Our current studies have focused on the application of ketene heterodimer β-lactones to the synthesis of deoxypropionate and dipropionate derivatives via catalytic hydrogenolysis/hydrogenation. Catalytic hydrogenolysis of the Z-isomer of ketene heterodimers provides access to anti-deoxypropionates with good to excellent diastereoselectivity (Org. Lett. 2015, 17, 3248-3251).
On the other hand, we have determined that catalytic hydrogenation of the E-isomer of ketene heterodimer provides access to syn,anti-β-lactones (dipropionate derivatives) bearing three stereogenic centers (Synthesis 2016, 2619). Preliminary results suggest that very good diastereoselectivity can be achieved (dr up to 49:1). A fully ‘reduced’ β-lactone of this type represents a dipropionate derivative, an important structural motif found in many promising chemotherapeutic agents (e.g. discodermolide) and antibiotics (e.g. erythromycin).
Other hydrometallation technologies (e.g. hydroboration) will be explored as a means of accessing all diastereomers of deoxypropionate and dipropionate derivatives with high diastereoselectivity (ideally dr >20:1). Once high diastereoselectivity has been achieved in elaborating ketene heterodimers to the requisite polyketide building blocks, the methods will be applied to the synthesis of interesting deoxypropionate natural products (e.g. mycocerosic acid and hexamethyldocosane).
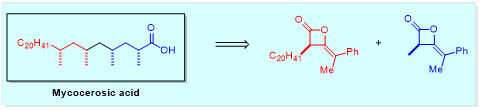
These natural products will then be investigated, through collaborations, as vaccines and as biomarkers for tuberculosis. In addition, the deoxypropionates will be converted to glycolipids for further biological evaluation (e.g. as immunomodulatory agents).
